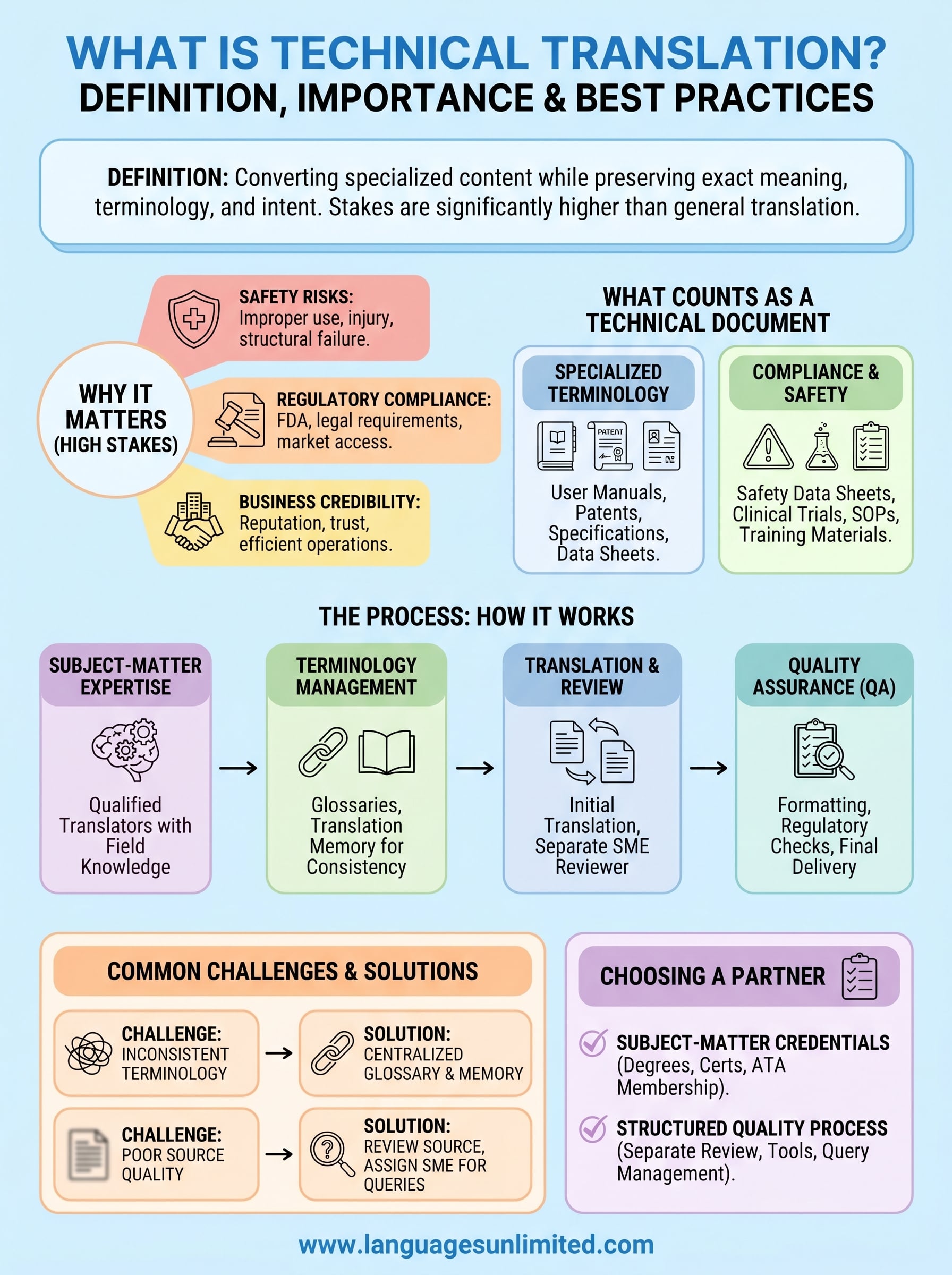
A user manual for an MRI machine. A patent filing for a new pharmaceutical compound. A safety data sheet for industrial chemicals. These documents share one thing in common: a single mistranslation can cause real harm. What is technical translation? It’s the process of converting specialized, subject-specific content from one language to another while preserving the exact meaning, terminology, and intent of the original. It goes well beyond general translation because the stakes, and the complexity, are significantly higher.
Technical translation covers documents in fields like engineering, medicine, IT, manufacturing, and law. These texts rely on precise terminology that often has no direct equivalent in the target language. A qualified technical translator doesn’t just speak two languages, they understand the subject matter deeply enough to make accurate decisions about every term, phrase, and instruction.
At Languages Unlimited, we’ve provided specialized translation services since 1994, working with legal, medical, and government clients across all 50 states. Our network of over ten thousand language professionals includes subject-matter experts who handle technical content daily. In this article, we break down what technical translation involves, the types of documents that require it, real-world examples, and the best practices that separate accurate technical translation from a costly mistake.
Why technical translation matters
When you understand what is technical translation, the next question is why it demands such a high level of care. The short answer: errors in technical documents carry real consequences. A mistranslation in a medical device manual can lead to improper use and patient injury. A flawed translation of a construction specification can result in structural failure. The precision these documents require isn’t optional; it’s built into the purpose of the content itself.
Accuracy has direct consequences
Technical documents inform decisions that affect safety, operations, and outcomes. When a factory worker follows a translated safety procedure, they trust that every instruction is accurate. When a patient reads a translated medication guide, they rely on the correct dosage and warning information being preserved exactly as written. A single substituted word or unclear instruction can create a chain of errors that’s difficult to reverse.
Getting technical translation right the first time protects people from harm that a correction issued weeks later cannot undo.
The financial cost of errors compounds quickly. Recalls, legal liability, regulatory fines, and contract disputes all become possible when translated content contains mistakes. For businesses operating across multiple markets, investing in qualified technical translation upfront is significantly less expensive than addressing the fallout from inaccurate documents after the fact.
Regulatory compliance depends on it
Industries like pharmaceuticals, medical devices, and aerospace operate under strict regulatory frameworks. In the United States, the FDA requires accurate labeling and documentation for products sold in or imported into the country. If your translated documents don’t meet those standards, your product won’t pass review either. Regulatory agencies in other markets hold equally high standards, and your translated content must reflect the specific requirements of each jurisdiction.
Your compliance documentation, product registrations, and safety data sheets often require certified translations that meet specific formatting and accuracy criteria. Working with translators who understand both the language and the regulatory environment keeps your organization on the right side of those requirements without costly revisions.
Your credibility follows your content
Sending a translated technical document to a client, partner, or regulatory body means that document represents your organization. Poorly translated content signals carelessness, and in technical fields, that impression can end business relationships before they gain traction. Clients in international markets expect the same level of precision and clarity in translated materials as they would in the original.
Strong technical translation also supports your internal teams. Engineers, technicians, and medical staff working in their native language are more efficient and make fewer errors when documentation is clear and accurately translated. The investment in quality technical content pays off across every team that depends on it to do their job correctly.
How technical translation works
Understanding how the process works in practice helps you set clear expectations before any project starts. Technical translation moves through several coordinated stages, from matching the right translator to your subject matter, through terminology management, and into structured quality review before final delivery.
Subject-matter expertise and terminology management
Selecting the right translator is the first and most critical step. A qualified technical translator brings both language fluency and working knowledge of your field, whether that’s industrial engineering, clinical medicine, or cybersecurity. Without that subject-matter depth, translated text can read grammatically correct while completely misrepresenting the technical meaning of the original.

Once the translator is in place, terminology management protects consistency across your entire document set. Project teams build glossaries that lock in approved translations for every key term, part name, and regulatory phrase. This ensures the same term reads identically across every file you send to clients, regulators, or manufacturing and distribution partners.
Review and quality assurance close the loop
After the initial translation is complete, a separate subject-matter reviewer checks the output against the source document. This step catches errors introduced under time pressure or from ambiguous source text, which even experienced translators encounter. A two-step review process is standard practice for any high-stakes technical content.
Separating the translator and reviewer roles is one of the most effective safeguards available in technical translation quality control.
The final stage of quality assurance checks formatting, numbering, and regulatory-specific requirements your documents must satisfy before reaching their intended audience. This includes verifying that figures, tables, and cross-references align correctly in the translated file, since layout errors in technical documents create just as much confusion as mistranslated terminology in a field or clinical setting.
What counts as a technical document
When you consider what is technical translation, it helps to understand exactly which documents fall into this category. Not every specialized text qualifies. Technical documents share a common trait: they contain precise, field-specific information that a general translator cannot accurately render without subject-matter knowledge.
Documents defined by specialized terminology
Some of the most common technical documents include manuals, specifications, and data sheets. These texts rely on standardized terminology that must match what engineers, technicians, or scientists use in their daily work. A product user manual for industrial equipment, for example, contains component names, operational sequences, and safety warnings that must translate without any deviation from the original meaning.
Patents and regulatory submissions also fall into this category. Patent applications require exact language to protect the scope of an invention, and any ambiguity introduced during translation can weaken legal protection in foreign markets. Regulatory submissions for medical devices or pharmaceuticals must satisfy jurisdiction-specific requirements, meaning the translator needs both language skill and regulatory awareness.
Technical content from fields like engineering, medicine, and law leaves no room for approximation; every term needs to carry exactly the weight the author intended.
Documents tied to compliance and safety
A second group of technical documents centers on safety and compliance obligations. Safety data sheets for hazardous materials must communicate risk accurately to workers in any language. Clinical trial documentation, informed consent forms, and medical protocols carry similar responsibilities; incorrect translations in these contexts directly affect patient safety and regulatory approval.

Training materials, standard operating procedures, and quality management documents also qualify as technical content. Your manufacturing teams or clinical staff rely on these materials to perform their roles correctly, and the consequences of unclear or inaccurate translations show up in operational errors, audit failures, and liability exposure.
Common challenges and how to avoid them
When you look closely at what is technical translation, several recurring problems appear that can compromise even well-planned projects. Knowing these challenges before they arise puts you in a position to address them proactively rather than after they’ve already created delays or errors across your final deliverables.
Inconsistent terminology across large projects
Technical translation projects spanning multiple documents or languages run into terminology inconsistency more often than any other single problem. When different translators handle separate files without shared reference materials, the same component can receive three different names across your documentation set. This confusion creates real operational problems for technicians, engineers, and clinical staff who depend on consistent terminology to perform their roles accurately.
The solution is direct: build a centralized glossary before translation begins and require all translators and reviewers to work from it throughout the project. A translation memory reinforces this by storing previously approved segments and flagging any deviation from established terminology, protecting consistency at scale.
Locking in approved terms before a project starts costs far less than correcting inconsistencies across thousands of words after delivery.
Poor source document quality
Your translation can only be as accurate as the source content you provide. Ambiguous phrasing, missing context, or inconsistent formatting in the original document forces translators to make judgment calls, and in technical fields, those judgment calls introduce risk. Undefined acronyms and unclear antecedents are two of the most common source-side problems that slow projects down and reduce accuracy in the final output.
Before sending documents for translation, review them for clarity at the sentence level. Assign a subject-matter expert from your team to answer translator queries promptly during the project. Clear source content combined with fast access to internal expertise shortens timelines and produces more reliable results without requiring costly revision rounds after delivery.
How to choose a technical translation partner
Knowing what is technical translation is only part of the picture. Choosing the right partner to handle your content determines whether your project succeeds or fails. Not every translation agency carries the expertise required for specialized content, so evaluating your options carefully before committing to a vendor protects both your timeline and your final deliverables.
Look for subject-matter credentials
Your translation partner should demonstrate verifiable expertise in your specific field, not just general language proficiency. Ask directly whether their translators hold relevant degrees, certifications, or professional backgrounds in areas like medicine, engineering, or law. A partner who can show you credentials that match your content type reduces the risk that technical terms get approximated rather than accurately rendered.
Membership in professional organizations like the American Translators Association signals that a provider operates under established quality and ethical standards. ATA membership requires translators to demonstrate language competence and commit to ongoing professional development, which supports the accuracy your technical documents require.
Verifying credentials before signing a contract takes minutes and prevents the kind of project failure that takes months to recover from.
Ask about their quality process
A reliable partner will walk you through their review and quality assurance workflow without hesitation. Confirm that a separate reviewer checks every translated file against the source, and that the partner uses translation memory and glossary management tools to protect consistency across all your documents.
Also ask about their process for handling translator queries when your source content is unclear. A partner with a structured query management system keeps projects moving without sacrificing accuracy. Confirm their turnaround capacity for your volume and whether they can scale if your project grows, so you are not switching vendors mid-project under deadline pressure.
Next steps
Understanding what is technical translation gives you a clear framework for approaching your next specialized project with confidence. The core takeaway is straightforward: accuracy, subject-matter expertise, and structured quality review are non-negotiable requirements for any technical document you need translated, whether it is a product manual, a regulatory submission, or clinical trial documentation.
Your next step is to evaluate your current translation needs against the criteria covered in this article. Identify the document types and subject areas involved, confirm whether your current vendor holds verifiable credentials in those fields, and check whether your source documents are clean and consistent before any project starts.
If you are ready to work with a team that has served legal, medical, and government clients since 1994 with a network of over ten thousand qualified language professionals, contact the Languages Unlimited team to discuss your project requirements.
